

The Fire Syringe demonstrates an adiabatic process. The autoignition temperature of cotton is 407oC (765oF).

A thin piece of pyrolytic graphite cuts through an ice cube. This material, of layered graphene, has excellent electron and thermal conductivity along the layers, but poor conductivity through the layers. This made it good heat shielding material for re-entry vehicles. It's diamagnetic properties are also anisotropic, with a very strong response to fields perpendicular to the layers.
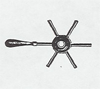
Identical small balls are attached by wax to the ends of six rods of different metals, radiating from a common metal center. The center of the rods is heated with a flame, and the small balls drop off at different times as the heat is conducted out by each different rod to melt the wax holding the ball.
Drinking Bird - The bird dips its beak into a beaker of water "taking a drink" and rotates back up. Cooling by evaporation from its beak draws up a colored liquid (methylene chloride) through a tube in its body overbalancing the bird so it takes another drink. This action repeats indefinitely.
Love Meter - Hold this device in your hand and heat from your hand boils methylene chloride causing it to flow through an intricate series of tubes.
See also Galileo's Air Thermometer [1]


How can you walk across hot coals?
Firewalking has been going on all over the world for thousands of years with written records going back to 1200 BC. Eastern Orthodox Christians in Bulgaria firewalk during popular religious feasts. So do Japanese Taoists, Buddhists, Indian Fakirs, the !Kung Bushmen, Polynesians, etc. Some claim that firewalking is an example of mind over matter, or a test of the protective power of faith.
It is true that the temperature of the coals is over 1000 degrees F (535 degrees C), and that human flesh burns at much lower temperatures, but temperature isn't the only part of the relevant physics. It wasn't until the 1770's that Joseph Black figured out the relation between thermal energy and temperature. (He later discovered Carbon Dioxide.) Different substances have different heat capacities. Water is the standard. It takes 4.18 Joules to raise the temperature of 1 cc of water 1 degree C. Our feet are mostly water. The coals have a much lower heat capacity than water. That means that the same amount of energy flowing away from the coals will lower their temperature much more than that same energy flowing to the feet will raise the foot's temperature. If the foot stays in contact with the coals, energy will keep flowing until they both reach the same temperature. However, this takes time, and how much depends on the heat conductivity. There are good heat conductors, like water, and poor conductors/heat insulators, such as ash. The feet cool down the local area of the coals they touch, and it takes time for energy to flow from the rest of the fire to the cool spot. You can sometimes see dull orange footprints in the coals right after someone walks. Water is a good heat conductor and energy transferred to the foot is rapidly conducted away from the contact points so the temperature doesn't rise to the burning point. Temperature, heat capacity, and thermal conductivity are all important in this demonstration.
A more familiar experience which involves the same physics is baking brownies in the oven set to 450 degrees F. Everything in the oven is 450 degrees, but you don't fear putting your hand in the oven air. The air has a very low heat capacity meaning it stores very little thermal energy. Air is also a heat insulator. Your hand (mostly water) cools the air locally and heats up very little. If you stick your finger in the brownie, you might get burned. It is mostly water like your hand and has a pretty good heat conductivity. Thermal energy will flow to your finger raising its temperature quickly. The metal pan is another matter. It has a high heat capacity and a high conductivity. Touch it without a potholder and you might instantly burn your fingers.
What the physics tells you, is that if you walk fast and don't stay in contact with the coals very long, you won't get badly burned. If you believe in mind over matter or the protective power of faith, then time shouldn't matter. This could be a life threatening delusion.
Even knowing the theory, firewalking is still dangerous in practice. There is a lot of energy in a glowing firepit at 1000 degrees F. Second degree burns in the form of blisters are common and more severe burns requiring a trip to the hospital have occurred. Sometimes a hot coal will stick to the foot causing a burn. There can also be hot spots in the fire, pieces of metal, or even pockets of hot steam locked up in the wood. Falling down in coals can be fatal. We will take care to make our walk as safe as possible.
Watch a video explanation of firewalking [2]. The video includes comments from Bernie Leikind, who got his start firewalking at UCLA.
One last note: Just because some aspects of firewalking and heat are "just physics", don't try to copy any fire stunt you might see. There have been many fire performers throughout history who used trickery to amaze audiences, and if you tried to duplicate their trick you would be severely injured. For instance, some performers scooped boiling lead into a ladle, and then poured it into their mouth. Shortly after, they spit out a chunk of cold lead with their teeth impressions in it. However, all was not as it seemed. The ladle had a hollow handle with mercury inside. Instead of scooping molten lead, mercury from the handle filled the ladle. Instead of pouring molten lead in their mouth, the mercury just went back into the handle. The cold lead with the teeth impressions had been hidden in the mouth beforehand.

In order to get liquid nitrogen we need a little extra notice. Some experiments are:
| Gas | In atmosphere | Boiling point | Melting |
| Nitrogen | 78.1% | 77 K, -196 C, -320 F | 63 K |
| Oxygen | 20.9% | 90 K, -183 C, -297 F | 54 K |
| Argon | 0.9% | 87 K, -186 C, -303 F | 84 K |
| Carbon Dioxide | 0.038% | 195 K,-78 C, -108 F | none |
| Helium | none | 4.2 K, -269 C, -452F | 0.95 K |
A flask of air connects at the bottom to a column of water. When the flask is heated by your hand, the air expands and the water falls. When it is cooled by evaporating alcohol, the air contracts and the water rises. Why doesn't this make a good thermometer? (It also responds to barometric pressure.)
What does the energy locked in 1 gram of sugar, 16 kilojoules, look like? Let's oxidize a gram and see.
This is an example of an exothermic reaction. The white powder is Potassium Chlorate. When melted it becomes a source of reactive oxygen. A typical gummy bear is 1 gram of sugar. Glucose + Oxygen ⇒ Carbon Dioxide + Water + energy
C6H12O6 + 6 O2 ⇒ 6 CO2 + 6 H2O + 16 kilojoules per gram of sugar
In cellular respiration sugars are oxidized and the resulting energy is stored in molecules of ATP. Each gram of sugar produces about 3.75 kilocalories or 16 kilojoules of food energy. The average human body utilizes about the same energy as a 100 watt light bulb. 100 watts x 24 hours x 3600 seconds/hour = 8,640 kilojoules per day. (Starvation is considered to be less than 1800 kilocalories or 6,830 kilojoules per day.) If we got all our energy from sugar and starch, we would need about 540 grams/day which is just over 1 pound per day.

If we could power our bodies with electricity from the utility it would take 2.4 kilowatt hours to operate at 100 watts for a day. At the rate of about 10 cents per kilowatt hour, the energy would cost 24 cents.
For more details on cellular respiration from the Khan Academy:
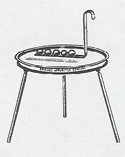
A set of five balls of different metals is heated in boiling water. The balls are then dropped onto a thin paraffin slab. They melt their way through at rates depending on their heat capacity (which depends on both the specific heat capacity of the metal and its mass).
Dissimilar metal wires with two junctions are hooked to a galvanometer. Heating one of the junctions with your fingers produces a substantial reading.
Links:
[1] https://demoweb.physics.ucla.edu/node/316
[2] http://www.youtube.com/watch?v=-W5FRl0qhOM
[3] https://demoweb.physics.ucla.edu/sites/default/files/demomanual/matter_and_thermodynamics/heat_and_temperature/ln2icecream.jpg