

A continuous cloud chamber shows tracks of charged particles. Advance notice is needed to obtain the dry ice necessary to operate the chamber. Thoron gas (thorium emination, Rn 220, half-life = 1 min.) can be blown into the chamber to produce alpha particle tracks. Since the daughter nucleus Po 216 with a half-life of 0.15 sec. is also an alpha-emitter, two pronged tracks will be seen in the chamber. A needle with Pb 210 also produces a-tracks. Two to five students look at this demonstration at once so it is best to arrange a little time at the beginning or end of class for them to come down and look.
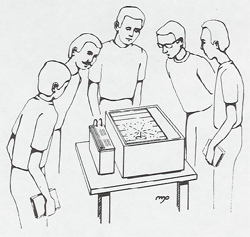
Cloud Chamber
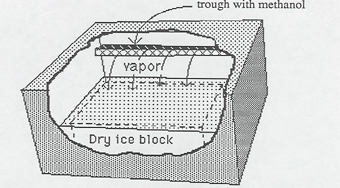
Methanol evaporates from the trough, and the vapor falls toward the cold dry ice (-100 F = -73 C). In the process the vapor is super cooled; that is, cooled below its normal condensation point. When a high speed charged particle from a radioactive source or from a cosmic ray passes through the super cooled vapor, it ionizes the air and methanol atoms along the way; i.e., it strips electrons from these atoms. These ions and electrons serve as condensation centers for the methanol vapor, which condenses out in tiny droplets along the track of the charged particle outlining its path.
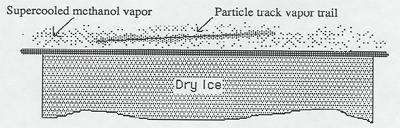
The charged particles from the radioactive source are typically helium nuclei (alpha particles). This source is "license free", meaning it is too weak to be considered dangerous by governmental regulatory agencies. Charged particles from cosmic rays are typically protons and muons.
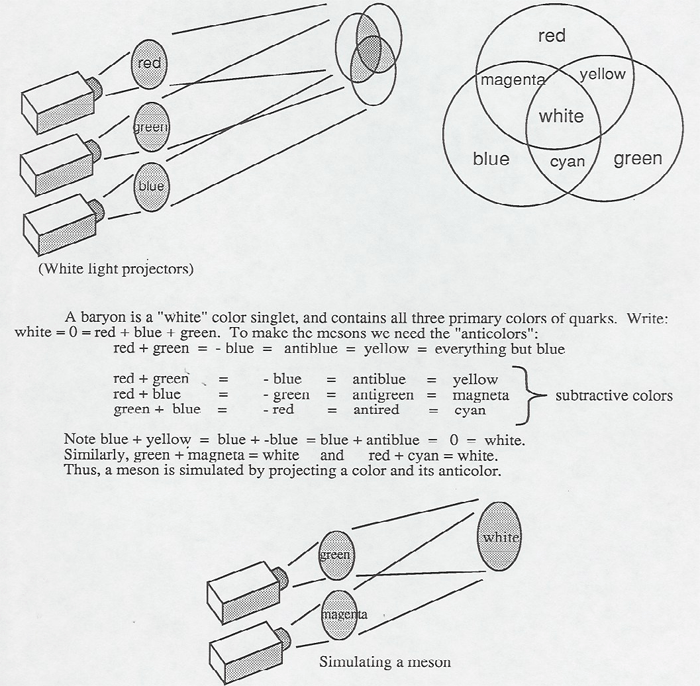
Using the color projector (Projected Colors [1]), you can simulate the quark structure of baryons and mesons.
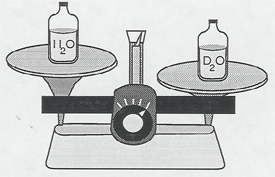
Heavy water, D2O, molecular weight 20, is about 10% heavier than ordinary water, H2O, molecular weight 18. Identically filled bottles of heavy water and tap water can be compared by hand or on a double pan balance. Of course, deuterium and heavy water are not radioactive.
The Physics Demontrations Group has a collection of videos on nuclear physics including:
The Physics Demontratons Group has a collection of transparencies on nuclear physics copied from Scientific American and other books including:
Small "license free" sources of activity < 0.1 microcurie can be taken into the class to activate a small hand held counter. Cloud chamber alpha sources of lead 210, beta source of strontium 90, thoriated tungsten welding rod, uranium glass (alpha beta gamma), Americium (alpha gamma) from a smoke detector and a hot fiesta ware cup and saucer with uranium glaze are available.
Americium-241, with a half-life of 432 years, is used in most domestic smoke detectors. Am-241 decays by emitting alpha particles and 60 keV gamma radiation to become neptunium-237.

Uranium glass was used to make a yellow tinted dinnerware from Victorian times. Sometimes called Vasoline glass, or depression glass, our bowl measures about 5 µSv/hr. You can tell uranium glass because it fluoresces green under blue or UV light.

Fiesta ware was the largest selling dish line in American history – 200 million dishes were shipped since 1936. The red/orange color glaze contains uranium. The government seized the company's uranium supply in 1943 out of fear it could be used to make a bomb. A single plate contains about 4.5 grams of uranium, mostly U-238. Production resumed in 1959 with depleted uranium (depleted of U-235) and continued until 1972 when it was discontinued out of concerns about uranium and lead leaching out of the glaze. Fiesta ware is the hottest source we have and measures over 100 µSv/hour at the plate surface, but this is not considered dangerous for display purposes. U-238 has a half-life of 4.5 billion years.
Radiation safety standards limit public exposure to 1 mSv/year and occupational exposure to 50 mSv/year. Natural background radiation is about 3 mSv/year.
Contrast that with a typical medical dose as shown below. This reading was taken about two hours after injection with Technetium-99m, a radioisotope used for heart imaging with a 6 hour half life.

A handheld Gieger-Mueller tube is available.
Links:
[1] https://demoweb.physics.ucla.edu/node/370