

This demonstration uses the microwave apparatus of experiment 2 in the 8D lab. The microwaves produced by a Gunn diode are picked up by a receiver and indicated on a meter visible to the class. When one of the 45 degree prisms is inserted into the beam, the meter reading drops to near zero - the microwaves have been totally internally reflected at right angles. You can demonstrate that this is the case by moving the receiver around to the right-angle position to pick them up. Now the receiver is returned to the original position, and a second prism is brought close to the first as shown above. When the gap is about one centimeter or less, the signal begins to increase - the microwaves have tunneled across the gap into the second prism. When the gap distance is reduced to zero, the signal reaches its full value. As the gap distance is increased, the signal drops off exponentially.
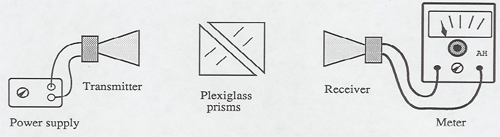
Remember, even electromagnetic microwaves are photons! This is a demonstration of particles penetrating a barrier.
The black body radiation curve can be demonstrated by using a radiation sensor hooked to a digital millivoltmeter. The carbon disulfide prism [1] is used to spread out the light of a slide projector lamp onto a screen . As you scan across the spectrum with the radiation sensor, the millivoltmeter shows the peak of the 3000 K tungsten filament in the infrared with the tails of the curve in the visible spectrum and further infrared.
The same demonstration can be done more qualitatively. Turn down the room lights and show the spectrum of "white" light on the wall. As you reduce the voltage to the lamp with the variac, the blue color dies away, and then the green, leaving only dull red of low intensity. (Of course, the 3000 K tungsten filament already peaks in the infrared so the initial "white" light is already quite red. Infrared itself can be demonstrated; see Infrared, Radiometer, and Maxwell's Spectrum [2])
|
The applet below shows the blackbody curve and colors corresponding to the given temperature. |
|||
|
When introducing the Bohr atom it is a good idea to review standing waves for the students, for example with Rudnick's String [4]. You can show how the string resonates for various harmonics as the driving frequency is changed.
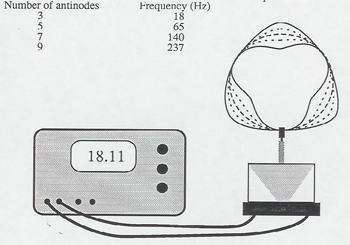
Another demonstration puts standing waves on a circular wire loop - standing waves around a circle. A mechanical wave driver controlled by a function generator shakes the loop at the bottom and it resonates with an odd number of antinodes on the circle at the frequencies:
| Number of antinodes | Frequency (Hz) |
| 3 | 18 |
| 5 | 65 |
| 7 | 140 |
| 9 | 237 |
The set-up below is an excellent demonstration of the wave nature of particles. An electron beam is passed through a carbon foil and the resulting diffraction rings displayed on the fluorescent screen of the tube. (Since the graphite crystals are randomly oriented, the diffraction pattern is rings.) From the accelerating voltage, DeBroglie's wavelength, and the diffraction ring diameter, you can calculate the atomic spacing of carbon.
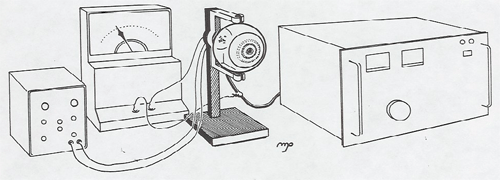
The students don't know what they are seeing. You take that into the class room and the students see green rings, but a physicist sees something astonishing, the wave nature of matter.
-Prof. George Igo
Students can see the spectral lines of hydrogen by looking at a arc tube on the lecture table through replica gratings. Several students can come down at once and look. You can also project the spectrum of mercury on a screen for the whole class to see (See Projected Mercury and Continuous Spectra [5]).
A simple demonstration of energy levels can be done with 4 LEDs, a handcranked generator, and a supercapacitor. As the capacitor is charged by the generator, first the red LED lights, then the green, then the blue. As the capacitor discharges, first the blue led goes dark, then the green, then the red. There is also a fourth IR LED which can't be seen by the eye, but can be seen on a cell phone camera or video camera when the room lights are out. It turns on first and turns off last. Each color has an energy (voltage) threshold which is related to the photon energy. The IR turns on at 1.2 V, red at 1.5 V, green at 2.0 V, and blue at 3.5 V.
The Frank-Hertz Experiment [6] shows atomic energy levels, but it is a very complicated demonstration.
Finally, a very simple demonstration of energy levels is fluorescent and phosphorescent materials with an ultraviolet light. The energetic UV light kicks the electrons up into high levels, and as they jump part way down immediately (fluorescent) or with some seconds of delay (phosphorescent with partially forbidden transitions), the electrons emit visible light of various colors. A green phosphor requires a blue or ultraviolet light to be activated. A red or green LED or laser won't make it glow, but a blue LED will.

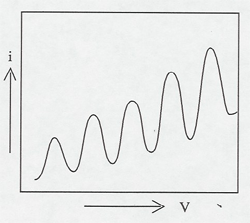
This classic 1914 experiment showed the existence of energy levels and their association with spectral lines. It involves an elaborate setup which requires over an hour of warm-up time and calibration. It is best suited for a lab experiment, or you could arrange to have it set up in a lab, and then bring the students in to watch it. Give plenty of extra notice.
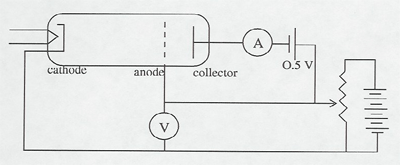
Electrons are accelerated in a nearly evacuated tube with a little mercury vapor in it from the cathode to anode by the voltage V. Those reaching the collector are retarded by 0.5 V. Thus, as V is increased from zero, there is no collector current at A until V > 0.5 V. Then the collector current rises until V reaches the excitation potential of a level in the gas atoms. The inelastic collisions reduce the electrons' energy to zero, and the current drops. As V is increased further, the current again increases until the electrons reach the energy of another level, or double that of the first level. The results can be seen on the current meter as the voltage is increased, or displayed on an oscilloscope screen using a sweep voltage.
A very simple demonstration of the photoelectric effect is performed with a zinc plate as the electrode of an electroscope. An ultraviolet lamp covered with glass is arranged to shine on the plate. The plate is charged negative with an electrophorus, and the electroscope needle diverges indicating the charge. The blue light of the lamp will not knock out electrons from zinc, but if the glass (opaque to UV) is removed from the lamp, the needle quickly falls as electrons are kicked away from the plate. The zinc plate must be cleaned with steel wool within an hour or so of the demonstration to remove the oxide.
A variation of this experiment has a spiral electrode with a positive voltage in front of the zinc plate with a sensitive current meter to measure the small current of the photoelectrons through the air.
The photoelectric effect is also done as experiment 4 in the 8E lab. The stopping voltage is measured as a function of wavelength (color) of the exciting light, and Planck's constant determined from the slope of the line.
The Ripple Tank [7] is useful to remind the students of wave interference. You might also wish to use the Acoustic Interference [8] demonstration with the ultrasonic transducers. With this setup, you can show that covering one of the two sources will increase the signal to the detector in the case of destructive interference, a key property of waves.
Light has wave properties: show interference with laser shining through slits (See Interference and Diffraction [9]).
Light has particle properties: show the Photoelectric Effect [10].
Electrons have wave properties: show Electron Diffraction [11].
The ratio of e/m for an electron can be measured with an apparatus consisting of a spherical evacuated electron tube mounted inside of a Helmholtz coil setup. The electrons are projected to circle in the magnetic field, and you measure the radius of the circle, the current to the Helmholtz coils, and the accelerating voltage to determine e/m. (See also E/M and Helical Electrons [12].) This is experiment 2 in the 8E lab.
Links:
[1] https://demoweb.physics.ucla.edu/node/208
[2] https://demoweb.physics.ucla.edu/node/207
[3] http://webphysics.davidson.edu/Applets/Applets.html
[4] https://demoweb.physics.ucla.edu/node/278
[5] https://demoweb.physics.ucla.edu/node/84
[6] https://demoweb.physics.ucla.edu/node/86
[7] https://demoweb.physics.ucla.edu/node/339
[8] https://demoweb.physics.ucla.edu/node/52
[9] https://demoweb.physics.ucla.edu/node/90
[10] https://demoweb.physics.ucla.edu/node/209
[11] https://demoweb.physics.ucla.edu/node/461
[12] https://demoweb.physics.ucla.edu/node/212